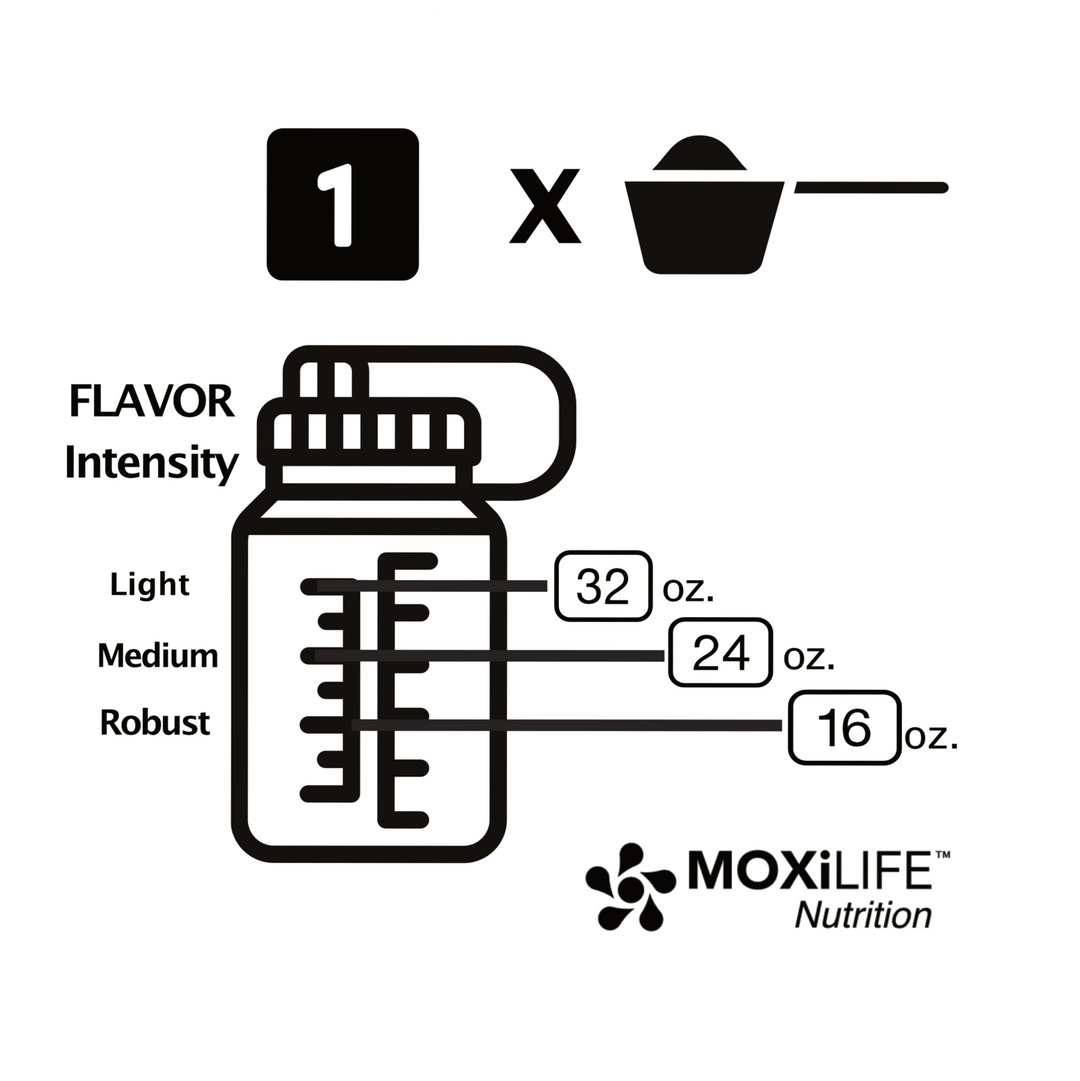
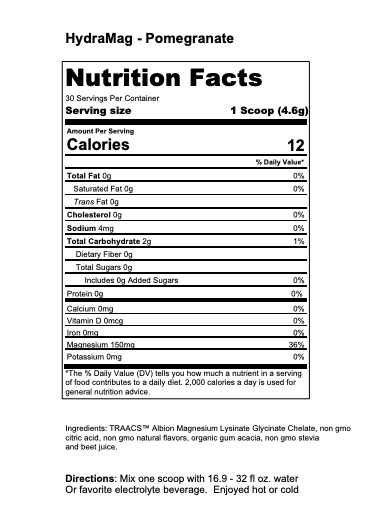
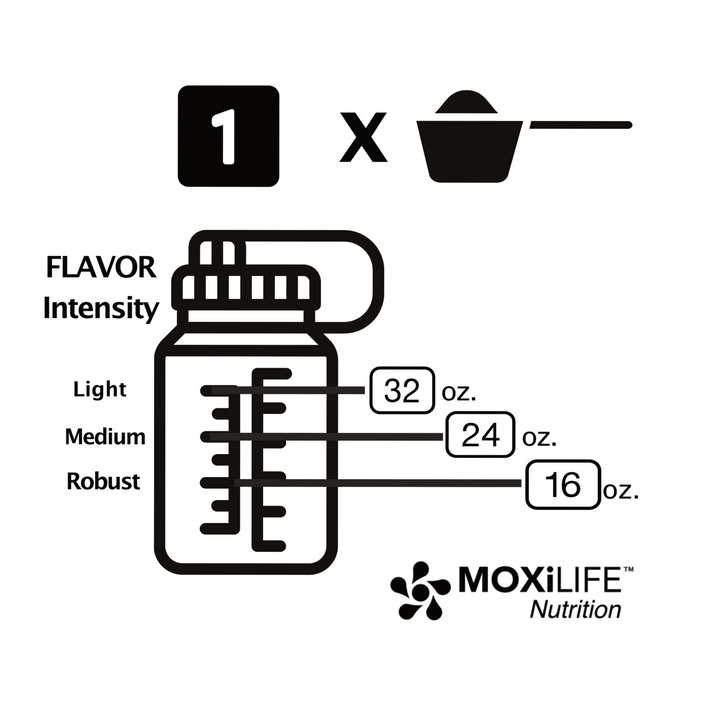
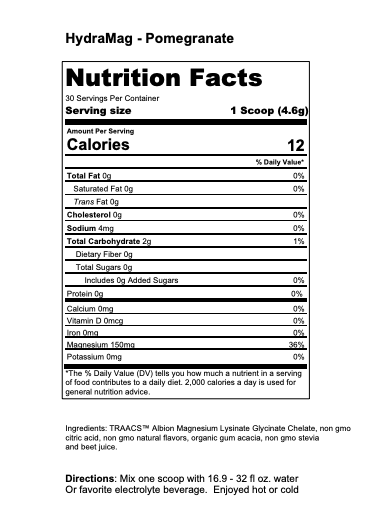
What are Mineral Differences and Can They Affect Athletic Performance?
What Are Minerals?

They are solid elements derived from nature. Minerals for nutritional purposes cannot be synthesized by the body. They must be utilized as natural elements from nature.
Why are minerals important?
Imbalances or deficiencies in minerals nutrition can affect the following systems:
- Immune…………………..Cu,Zn,Fe,Se
- Energy Production………Mg, P, Mn
- Hormone System………..Fe,Mn, Zn, Cu, Mg, K
- Vitamin Production………Co
- Blood Production…………Cu, Fe
- Enzyme Systems………….Zn,Cu, K, Mg, Mn,B, P
- Skeletal System…………..Ca, Mg ,Zn, Mn, B, P
- Reproductions …………….P, Cu, K, Mn, Zn, Mg
What is an Endurance Mineral Chelate?
Organic ring-shaped molecules made of minerals that are chemically bonded to amino acids. This type of mineral structure is the same one found naturally and in whole foods. Amino acid chelated minerals are up to 4x more bioavailable than mineral salts (‘inorganic’ minerals bonded with salts), which are the most common type of minerals found in mineral supplements today.
A mineral chelate; i.e. Zinc bisglycinate, Magnesium Lysinate Glycinate, Calcium bisglycinate etc. is: stable, fully reacted; and surrounded or ‘enveloped’ by amino acids. The process of internal chelation is completed with the expenditure of our bodies energy to bond the amino acid and mineral. This creates an efficient pathway for our bodies to transport organic minerals across the intestinal wall, as a part of digestion.
TRAACS® minerals are prepared to closely duplicate the natural chelation process which occurs in the body. TRAACS® minerals start as inorganic rocks and are converted into organic minerals.
These are the perfect absorbable form of minerals because of the:
- molecular design
- the added amino acids
- increased efficiency of absorption and
- reduced energy required to make them absorbable forms.
Why use amino acids?
- Amino acids are easily recognized and absorbed by the body. Dipeptide and single amino acids are the first inline for intestinal uptake after digestion.
- Dipeptides are faster in absorption than single amino acids.
- Minerals bonded/chelated to amino acids in a ‘dipeptide -like’ structures, allows the minerals to be absorbed in this active transport system.
Why is it important to have a stable (covalent) bond?
If the bond is not stable; the structure will loose integrity during digestion and compromise availability through the intestinal walls.If it falls apart it returns to an ionic form which forces the body to make attempts at restructuring the bond with amino acids found in food sources.
What makes TRAACS™ chelates so biologically effective?
They are manufactured to <800 Daltons = nano sized. (fun sized)
In order for the molecule to pass through the intestinal wall it has to be the optimal size; which is measured in Daltons.
The intestinal wall is much like a filter, to large it will continue down the waste management pathway.
Your body does not need to expend energy converting inorganic(ionic) minerals such as Oxides and Carbonates into organically recognized forms:
If the mineral is not an organic mineral chelate the inorganic mineral will need to utilize your body's energy to convert the inorganic mineral into a ‘dipeptide-like’ and recognizable form.
Mineral Chelates are Electrically Neutral:
As a mineral chelate: the electrons are tied up within the bond between the mineral and amino acid.The ‘free electrons’ which we find with inorganic, electrically positive or negative, are what causes interactions with other dietary components, rendering the mineral potentially un absorbable.
Electrically charged minerals can interact with compounds in our natural food sources; i.e. Phytates, oxylates, tannins, which reduces absorption considerably.
In addition to grabbing on to other dietary components these have the potential to deactivate and reduce active nutrients and medications efficacy:
- Vitamin E
- ascorbic acid
- various B vitamins
- Rx medications